"Enzymes are produced according to instructions encoded in DNA" – if I had told this to a professor of biology a hundred years ago, he would probably have thought I'd slipped on a Petri dish and hit my head. Today it appears in all biology textbooks. But who coined it? How and when was this conclusion reached in the first place? Let's discover how two scientists first showed that mutations can affect biochemical processes inside cells.
Advertisement
This article was inspired by the course “Bacterial Genetics”, taught by Prof. Eyal Gur from Ben-Gurion University.
Our genetic code contains the instructions for producing proteins in the cell. DNA sequences* (genes) are transcribed into RNA molecules, that leave the nucleus and are translated into proteins [1]. A change in the DNA sequence (a mutation) may alter the structure of the resulting protein. Proteins translated according to “faulty” instructions may fail to function properly and can sometimes cause disease. In the era of modern biology, the connection between mutations and heredity on the one hand, and protein activity or disease on the other, is obvious. But when and how was this link discovered?
Until the beginning of the 20th century, protein research was part of biochemistry. This field, among other things, investigates the activity of enzymes. Enzymes are proteins that act as molecular machines, accelerating chemical reactions in the cell. Enzymes function like workers on an assembly line: at one end lies a starting material, at the other end a product. The metabolism of living organisms is based on pathways of reactions catalyzed by different enzymes. Enzymes provide the organism the ability to convert substances from the environment, or from within the body, into new molecules required for life. Genetics, on the other hand, focused mainly on the inheritance of traits. The connection between these two disciplines was discovered only in 1902. British physician Archibald Garrod observed that alkaptonuria (“black-urine disease,” in which a compound accumulates in the urine and oxidizes to black upon exposure to air) follows a recessive Mendelian inheritance pattern. In other words, if both parents carry the disease, one quarter of their children will be affected [2]. In short, diseases characterized by a metabolic defect can be hereditary!
Garrod’s observation did not cause a stir, but about 30 years later, in 1935, two scientists, George Beadle and Boris Ephrussi, were intrigued by his ideas and decided to test whether a link between genes and protein activity could be proven [3]. Their research tool was the fruit fly (Drosophila melanogaster). They matched traits of individual flies, like eye color, with specific genes in the DNA. This way, they assigned each genetic mutation to a particular trait (phenotype). During the early developmental stage of fruit flies, their larva body is divided into sections that develop into different organs in the adult fly. A section can be removed or replaced with another, and the change will manifest in the adult. For instance, if a section destined to become an eye is transplanted into the larval abdomen segment, an adult fly will develop with an eye in its abdomen (yuck).
The researchers had different mutant flies with defects in eye color. Each mutant showed a different hue. Flies with “normal” eye color are called wild type. When studying the inheritance pattern of a mutation, we cross flies and count how many offspring express the mutation. But when we transplant tissue from a mutant fly (for example, eye tissue) into a wild-type fly, what will be the color of this eye in the adult fly? Because this is not a cross between a male and a female, the wild-type eye-color trait cannot simply be passed to the mutant eye… Or can it?
This claim is true for most of the mutants, except for two: Vermillion (vm) and Cinnabar (cm). Transplanting eye tissue from larvae of these mutants into a wild-type larva produced a fly with the normal, wild-type eye color (see Illustration 1).
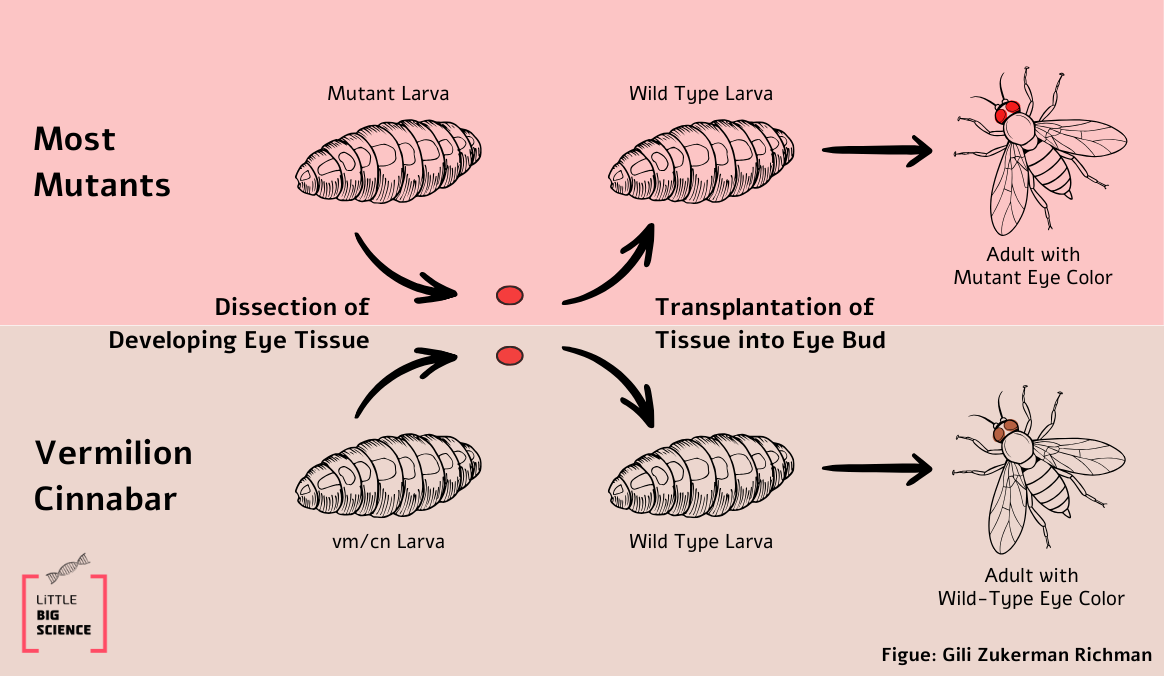
Illustration 1.
In this experiment, the adult fly was not produced by crossing, but by transplanting a mutant eye tissue into a wild type body. This means that there was no mixing of the mutant DNA with that of the wild type. So how can this result be explained? The researchers hypothesized that the wild-type body supplies the mutant eye with the missing intermediate component that is required to complete the synthesis of the eye pigment (call it i, see Illustration 2). If so, this result indicates that mutant flies have a defective enzyme in the biochemical pathway of eye pigment production.
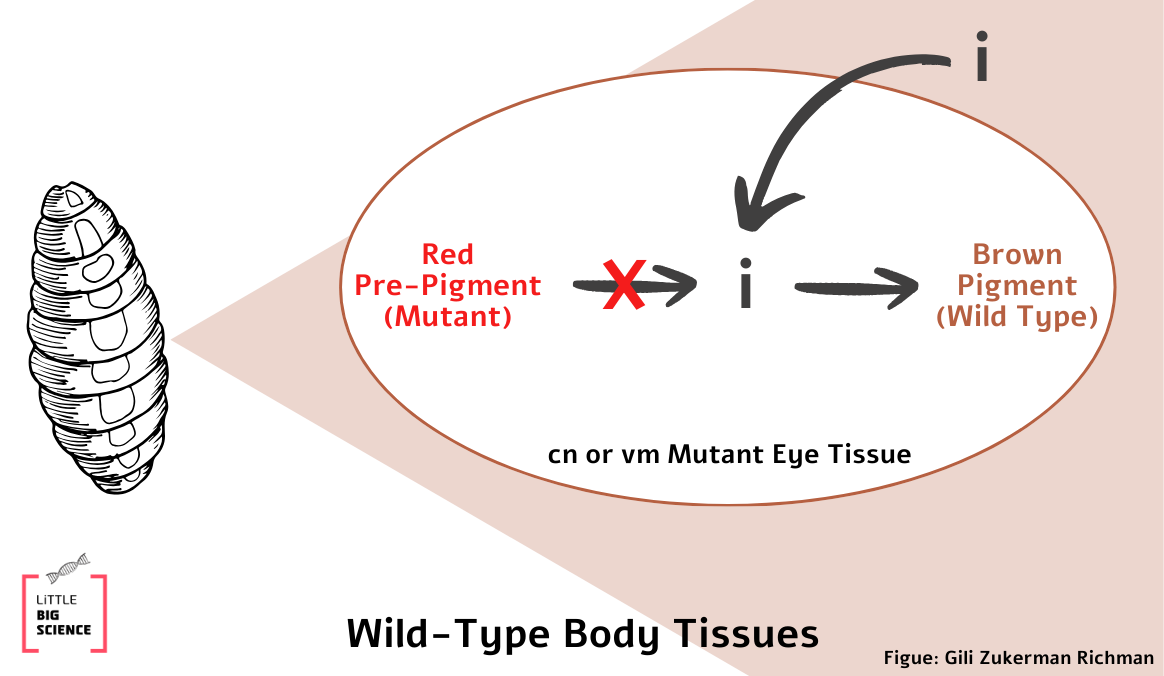
Illustration 2.
This raises another question: are both cn and vm mutants defective in the same enzyme? The researchers performed a complementation test to answer this. If the two mutants are defective in the same enzyme, neither can produce the intermediate i that is required for normal eye color. Therefore, if eye tissue from one mutant is transplanted into the other mutant, the mutant body tissues cannot provide the eye tissue with the metabolic components it lacks (in contrast to when mutant tissue is transplanted into a wild-type body). But if the mutants are defective in different enzymes, each is “stuck” with a different intermediate (call them i₁ and i₂) at different steps in the pathway. Remember: an enzyme that is non-functional in one mutant may work normally in the other! In that case, one mutant can “rescue” the other by providing the missing intermediate.
What was the result of the experiment?
When the researchers transplanted eye tissue from a cn mutant into the body of a vm mutant, the adult fly had the eye color of cn. This meant that the vm mutant could not supply the missing component to cn.
In the reciprocal experiment, when eye tissue from a vm mutant was transplanted into a cn body, the adult fly emerged with wild-type eye color! This meant that the cn mutant supplied the vm eye with the missing component. Based in these findings, the scientists concluded not only that the two genes encode different enzymes, but also that proper activity of one enzyme depends on the proper activity of the other, so that completion of the eye-color pathway is only possible when both enzymes function correctly (Illustration 3).
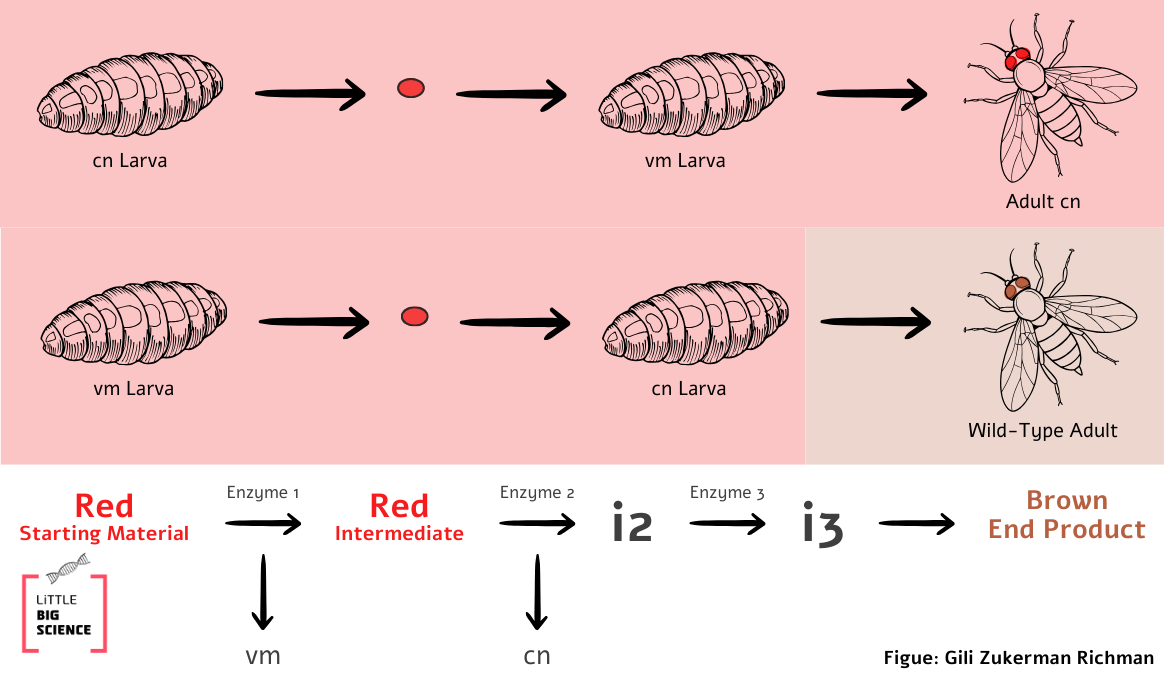
Illustration 3.
Following these experiments, the researchers coined the term epistasis. This is a circumstance where the expression of one gene is masked by the expression of one or more other genes. This was the first evidence that different genes are responsible for producing different proteins, and that distinct genes can act within the same metabolic pathway.
*Who said that “genes” reside in DNA? Feel free to read the posts “What is the hereditary material?” [4] and “From material to trait” [5].
Hebrew editing: Smadar Raban
English editing: Gloria Volohonsky
Sources and further reading