The aurora, including the famous northern lights (aurora borealis) and the southern lights (aurora australis), is one of nature’s most mesmerizing phenomena. Observing this magnificent sight requires a precise convergence of conditions, each explained by a plethora of physical principles: From quantum mechanics and electromagnetism to thermodynamics and astrophysics. Hold on tight, we are taking a deep dive.
Advertisement
Other than light and heat, electrically charged particles, such as electrons, are expelled from the Sun’s corona, its outer layer, and race through space at high speeds in every direction. This stream of particles is called the “solar wind”, and it constantly reaches Earth.
If the solar wind were to hit our atmosphere directly, the wind's charged particles would collide with it and gradually strip it away—just as happened to Mars billions of years ago [1]. Our protection lies in Earth’s magnetic field. The field is sustained by a self-reinforcing process explained by dynamo theory: In the outer core of Earth, a molten, electrically conducting mixture of iron and nickel circulates. Heat released from the inner core drives convection, so that hotter material rises while relatively cooler material sinks. Earth’s rotation deflects these flows via the Coriolis force [2]. The movement of the conducting fluid generates electric currents, and according to the laws of electromagnetism, an electric current produces a magnetic field.
Earth therefore behaves like a giant magnet whose field lines loop outward and, near the poles, plunge back into the atmosphere, acting as a natural funnel for charged particles. The magnetic field carried by the solar wind changes constantly because of the Sun’s rotation and coronal mass ejections [3]. When this interplanetary field is oriented opposite to Earth’s field, a process called magnetic reconnection [4] occurs. Energy is then transferred into Earth’s magnetosphere, accelerating electrons toward the upper atmosphere. Collisions between these electrons and atmospheric components produce the aurora (details on how the colors are produced to follow). The more powerful the solar eruptions, the stronger the aurora, allowing it to reach closer to Earth’s mid-latitudes.
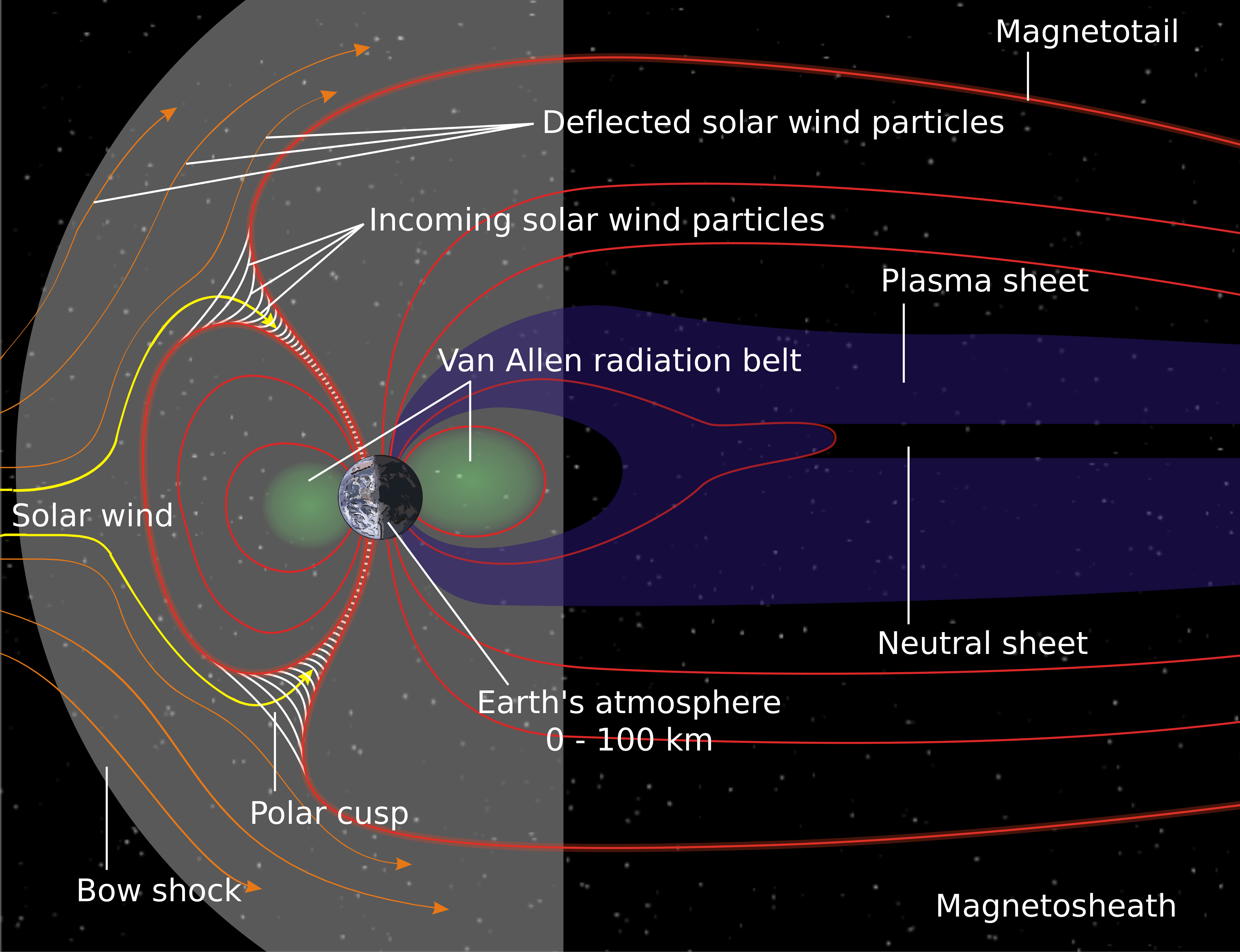
Figure 1: Schematic illustration of the solar wind (left) interacting with Earth’s magnetosphere—the region dominated by Earth’s magnetic influence. Earth’s magnetic field lines are drawn in red. Source: NASA.
Although the aurora poses no danger to ground-based observers because of its great height, the magnetic storms that create it can harm sensitive technologies. Rapidly changing magnetic fields induce unwanted electric currents in conductors. These currents disrupt radio communication, degrade the accuracy of satellite-based navigation systems, and in extreme cases damage power grids. The most famous example is the Quebec blackout of 1989: an intense magnetic storm overloaded the grid, prompting automatic protection systems to shut it down, leaving millions without electricity for nine hours [5].
And now for the artistic part—the physics behind the colors. When energetic electrons collide with atoms and molecules in the atmosphere, they transfer energy that can push those particles to excited quantum states with higher energy than their “normal” ground state. As the excited states decay to lower energies, photons of specific wavelengths, i.e., auroral colors, are emitted. Each color arises from a particular combination of altitude and atmospheric composition:
💚Green: The most common auroral color, appearing at altitudes of roughly 100–200 km. It occurs when an oxygen atom transitions from a higher to a lower excited state, emitting a green photon. The atom remains in a metastable state that would normally decay by emitting red light as well, but at these relatively dense altitudes collisions with nitrogen and oxygen molecules dissipate the excess energy as heat before the red photon can be emitted.
❤️Red: Occurs above about 200 km. The emission arises when an excited oxygen atom returns directly to its ground state and releases a red photon. At these heights the atmosphere is thin enough that the atom can radiate before colliding with another particle.
💙Blue: Appears at altitudes of roughly 100–120 km. It is produced when an energetic electron strikes a nitrogen molecule and ejects one of its electrons, ionizing it. The ionized, excited molecule then emits a blue photon. Although blue and green are generated at similar heights, green is more prevalent because exciting an oxygen atom requires less energy than ionizing and exciting a nitrogen molecule—the latter demands not only excitation energy but also the ionization energy needed to remove an electron.
💜Purple: Seen at lower altitudes, below 100 km, during particularly intense auroral events when very energetic particles penetrate deeper into the atmosphere. Emissions from ionized nitrogen produce blue light, while emissions from neutral nitrogen over a broader wavelength range yield pinkish hues. The combination appears purple to our eyes.
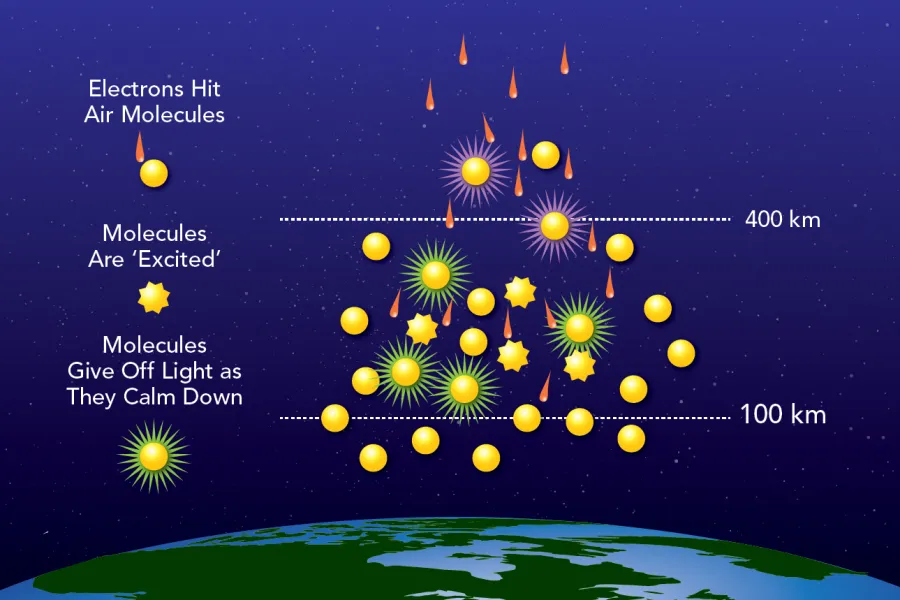
Figure 2: A conceptual illustration of the reaction that creates the aurora—electrons interact with atmospheric oxygen and nitrogen, transferring energy that excites them. The characteristic auroral light is emitted as the particles return to lower energy states. The figure shows how different colors are emitted at different altitudes: red originates higher than green, as explained in the text. Source: NASA.
In summary—the aurora is real-time, altitude-dependent spectroscopy!
To maximize your chances of seeing it, be near the poles, under dark, clear skies, during strong solar eruptions. In other words, don’t forget to pack a dash of luck.
Now that you’ve read the guide—enjoy the show, so long and thanks for all the fish.
Hebrew editing: Shir Rosenblum-Man
English editing: Elee Shimshoni
Sources
- How the solar wind stripped most of Mars’s atmosphere
- What is the Coriolis effect, NOAA
- Coronal mass ejection, Wikipedia
- Explanation and simulation of magnetic reconnection, NASA
- The 1989 Quebec geomagnetic storm,Wikipedia
- Whiter, D. K., Partamies, N., Gustavsson, B., & Kauristie, K. (2023, January). The altitude of green OI 557.7 nm and blue N2+ 427.8 nm aurora. In Annales Geophysicae (Vol. 41, No. 1, pp. 1–12). Göttingen, Germany: Copernicus Publications.
- Rees, M. H. (1989). Physics and Chemistry of the Upper Atmosphere. Cambridge University Press. Open access in English.